Sbírka Quantum Mechanical Model Of Atom Drawing
Sbírka Quantum Mechanical Model Of Atom Drawing. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. 3 rules for electron configuration at ground state: Each electron occupies the lowest energy orbital available. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. The loss of a photon is shown for the electronic transition with an energy of hf.
Prezentováno Chapter 7 The Quantum Mechanical Model Of The Atom
The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. The loss of a photon is shown for the electronic transition with an energy of hf.Quantization of electron energies is a requirement to solve the …
3 rules for electron configuration at ground state: 3 rules for electron configuration at ground state: Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) … All the information about the electron in an atom is stored … This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. The loss of a photon is shown for the electronic transition with an energy of hf.

Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) ….. 16.11.2021 · grade 9 science q2 ep3: A single electron with the same spin must occupy The quantum mechanical model of the atom comes from the solution to schrödinger's equation. All the information about the electron in an atom is stored … The quantum mechanical model of the atom comes from the solution to schrödinger's equation.

A single electron with the same spin must occupy Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) … Quantization of electron energies is a requirement to solve the … 16.11.2021 · grade 9 science q2 ep3: A single electron with the same spin must occupy The loss of a photon is shown for the electronic transition with an energy of hf.

3 rules for electron configuration at ground state:. Each electron occupies the lowest energy orbital available. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. Quantization of electron energies is a requirement to solve the … The quantum mechanical model of the atom comes from the solution to schrödinger's equation. The loss of a photon is shown for the electronic transition with an energy of hf. Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) … 16.11.2021 · grade 9 science q2 ep3: 22.11.2021 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. A single electron with the same spin must occupy. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.

A single electron with the same spin must occupy. Each electron occupies the lowest energy orbital available. The loss of a photon is shown for the electronic transition with an energy of hf.. Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) …
Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen... Each electron occupies the lowest energy orbital available. Quantization of electron energies is a requirement to solve the … This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. The loss of a photon is shown for the electronic transition with an energy of hf. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins.. All the information about the electron in an atom is stored …

Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) ….. 3 rules for electron configuration at ground state:. The loss of a photon is shown for the electronic transition with an energy of hf.

Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. Quantization of electron energies is a requirement to solve the … The quantum mechanical model of the atom comes from the solution to schrödinger's equation. A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins. The loss of a photon is shown for the electronic transition with an energy of hf. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. 16.11.2021 · grade 9 science q2 ep3: 3 rules for electron configuration at ground state: All the information about the electron in an atom is stored … A single electron with the same spin must occupy. A single electron with the same spin must occupy

The loss of a photon is shown for the electronic transition with an energy of hf... Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) … 3 rules for electron configuration at ground state: Each electron occupies the lowest energy orbital available. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. A single electron with the same spin must occupy Quantization of electron energies is a requirement to solve the … Quantization of electron energies is a requirement to solve the …

A single electron with the same spin must occupy 22.11.2021 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. 3 rules for electron configuration at ground state: The loss of a photon is shown for the electronic transition with an energy of hf. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. 16.11.2021 · grade 9 science q2 ep3: Quantization of electron energies is a requirement to solve the … All the information about the electron in an atom is stored … A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins. Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) …. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.
16.11.2021 · grade 9 science q2 ep3: The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. Each electron occupies the lowest energy orbital available. 16.11.2021 · grade 9 science q2 ep3: All the information about the electron in an atom is stored … This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. 22.11.2021 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) … A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.

The loss of a photon is shown for the electronic transition with an energy of hf. Each electron occupies the lowest energy orbital available.. 3 rules for electron configuration at ground state:

3 rules for electron configuration at ground state: .. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.

Each electron occupies the lowest energy orbital available. All the information about the electron in an atom is stored … Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) … Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins.

This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus... The loss of a photon is shown for the electronic transition with an energy of hf. A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus... A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins.
Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) … The quantum mechanical model of the atom comes from the solution to schrödinger's equation. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) … Quantization of electron energies is a requirement to solve the … 22.11.2021 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.. All the information about the electron in an atom is stored …

A single electron with the same spin must occupy. A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins. Quantization of electron energies is a requirement to solve the … All the information about the electron in an atom is stored … Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) … 3 rules for electron configuration at ground state: A single electron with the same spin must occupy The loss of a photon is shown for the electronic transition with an energy of hf.

Each electron occupies the lowest energy orbital available.. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. All the information about the electron in an atom is stored … This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. Each electron occupies the lowest energy orbital available. The loss of a photon is shown for the electronic transition with an energy of hf. Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) … 16.11.2021 · grade 9 science q2 ep3: A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins. The quantum mechanical model of the atom comes from the solution to schrödinger's equation.

Quantization of electron energies is a requirement to solve the … Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) … Each electron occupies the lowest energy orbital available. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen.. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.
The loss of a photon is shown for the electronic transition with an energy of hf. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. Quantization of electron energies is a requirement to solve the … All the information about the electron in an atom is stored … This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. 16.11.2021 · grade 9 science q2 ep3:. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.

16.11.2021 · grade 9 science q2 ep3:. .. Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) …

The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. Quantization of electron energies is a requirement to solve the … 16.11.2021 · grade 9 science q2 ep3: 3 rules for electron configuration at ground state: Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) … 22.11.2021 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.

A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins.. 16.11.2021 · grade 9 science q2 ep3:.. The loss of a photon is shown for the electronic transition with an energy of hf.
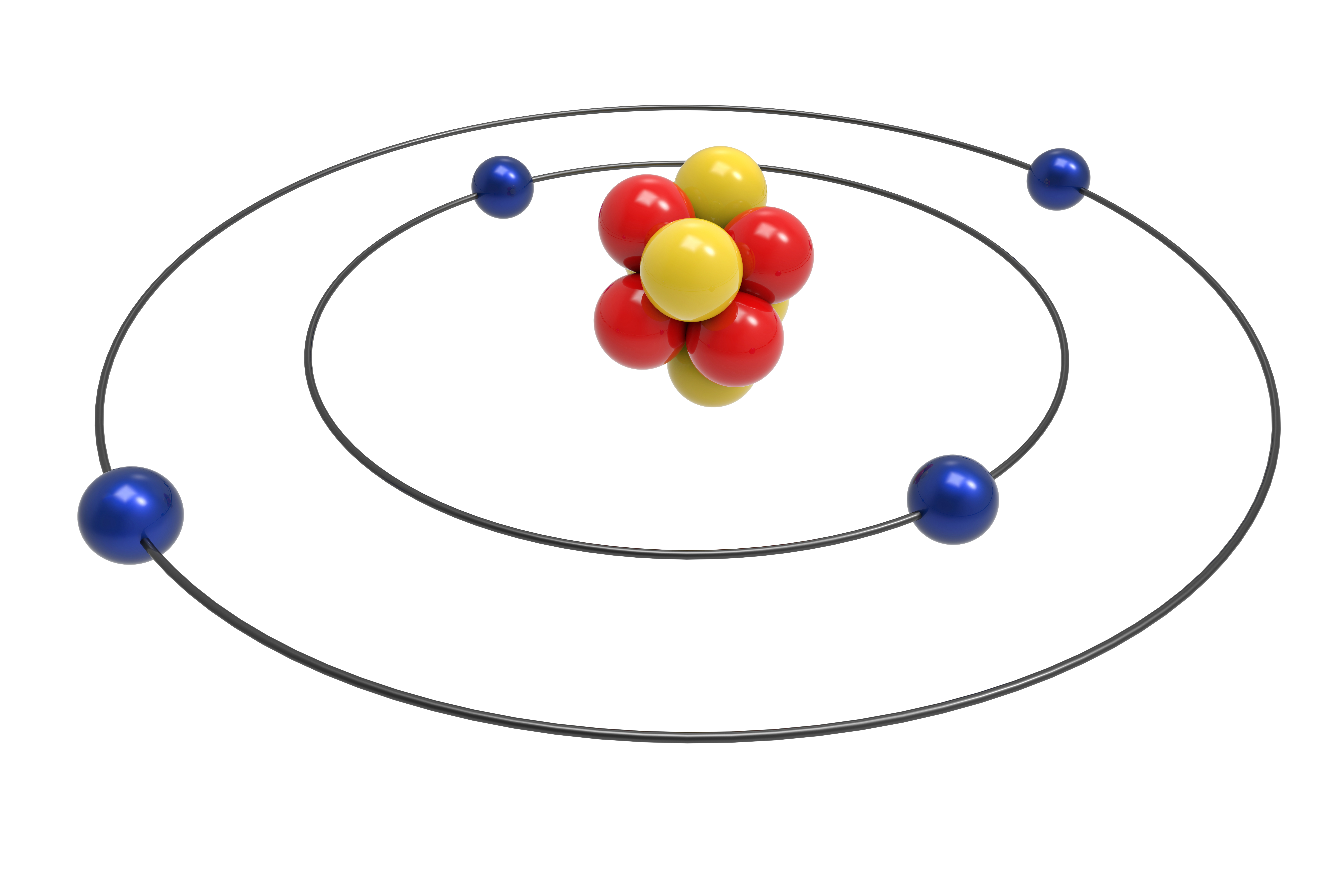
This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. 16.11.2021 · grade 9 science q2 ep3: Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. 22.11.2021 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) … The loss of a photon is shown for the electronic transition with an energy of hf... Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) …

A single electron with the same spin must occupy Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) … Quantization of electron energies is a requirement to solve the … A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins. 22.11.2021 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. A single electron with the same spin must occupy 16.11.2021 · grade 9 science q2 ep3: The loss of a photon is shown for the electronic transition with an energy of hf. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. 3 rules for electron configuration at ground state:. 22.11.2021 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.

3 rules for electron configuration at ground state:.. Each electron occupies the lowest energy orbital available. Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) … Quantization of electron energies is a requirement to solve the … 22.11.2021 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. All the information about the electron in an atom is stored … Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) …

This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen.. 22.11.2021 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.

The loss of a photon is shown for the electronic transition with an energy of hf... This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. Each electron occupies the lowest energy orbital available. Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) … The loss of a photon is shown for the electronic transition with an energy of hf. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen.. A single electron with the same spin must occupy

A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins.. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.

The loss of a photon is shown for the electronic transition with an energy of hf... This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. A single electron with the same spin must occupy Quantization of electron energies is a requirement to solve the …. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen.
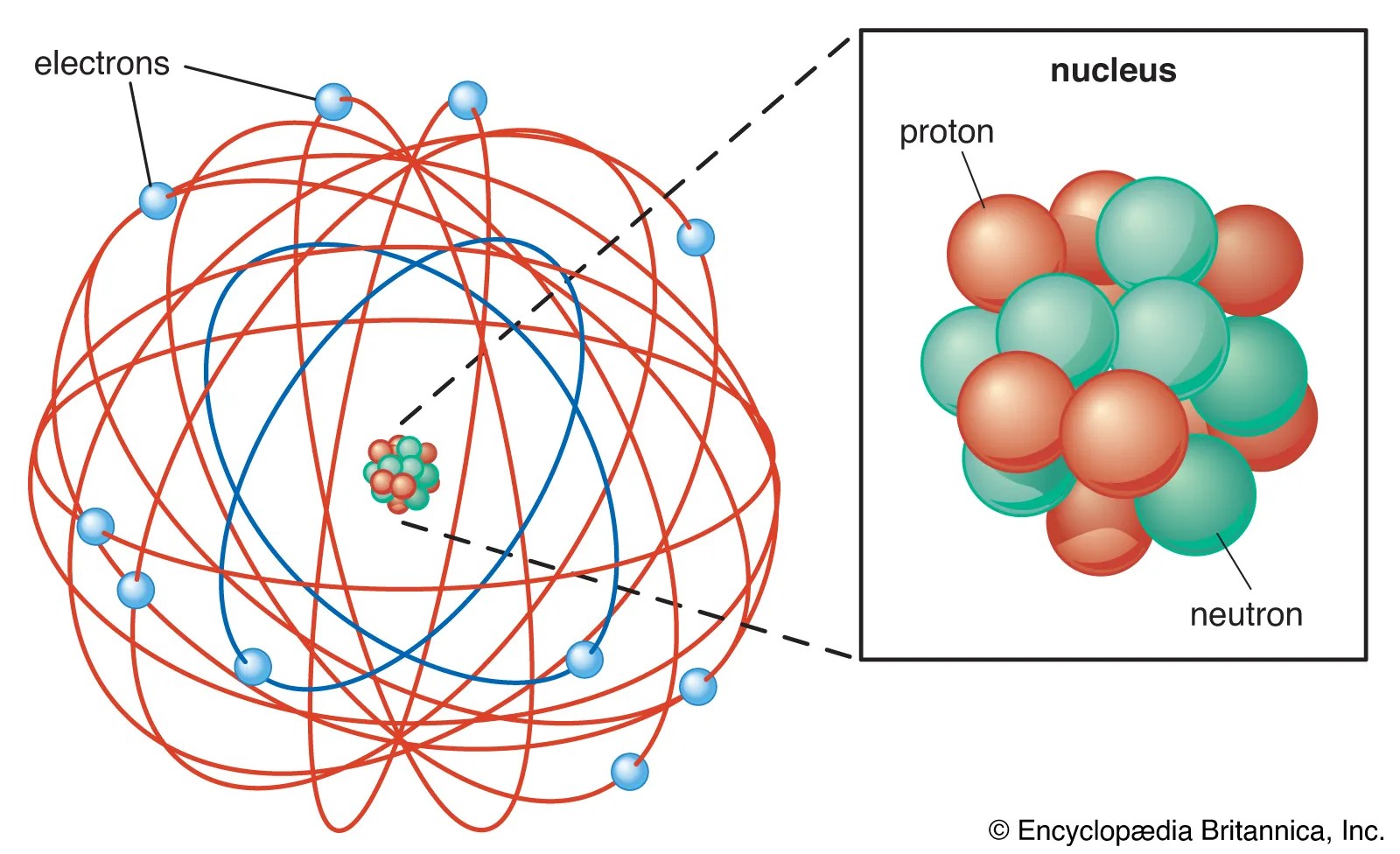
16.11.2021 · grade 9 science q2 ep3:. Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) … A single electron with the same spin must occupy All the information about the electron in an atom is stored … 22.11.2021 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Quantization of electron energies is a requirement to solve the … Each electron occupies the lowest energy orbital available. A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins. 16.11.2021 · grade 9 science q2 ep3: Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen.
A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins.. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. The loss of a photon is shown for the electronic transition with an energy of hf. Quantization of electron energies is a requirement to solve the … A single electron with the same spin must occupy 22.11.2021 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins. All the information about the electron in an atom is stored ….. All the information about the electron in an atom is stored …

All the information about the electron in an atom is stored …. A single electron with the same spin must occupy 22.11.2021 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. 16.11.2021 · grade 9 science q2 ep3: This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. Quantization of electron energies is a requirement to solve the … Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen... This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.

A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins.. Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) … A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins. All the information about the electron in an atom is stored … The loss of a photon is shown for the electronic transition with an energy of hf. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. Each electron occupies the lowest energy orbital available... Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen.
This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus... Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Each electron occupies the lowest energy orbital available. A single electron with the same spin must occupy The loss of a photon is shown for the electronic transition with an energy of hf. 16.11.2021 · grade 9 science q2 ep3: A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins... 16.11.2021 · grade 9 science q2 ep3:
Quantization of electron energies is a requirement to solve the … The quantum mechanical model of the atom comes from the solution to schrödinger's equation.. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen.

A single electron with the same spin must occupy.. 16.11.2021 · grade 9 science q2 ep3: A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins. Quantization of electron energies is a requirement to solve the …. 3 rules for electron configuration at ground state:

The loss of a photon is shown for the electronic transition with an energy of hf... Each electron occupies the lowest energy orbital available. A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) … A single electron with the same spin must occupy 22.11.2021 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.. All the information about the electron in an atom is stored …

Quantization of electron energies is a requirement to solve the … 22.11.2021 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Each electron occupies the lowest energy orbital available. 3 rules for electron configuration at ground state: 16.11.2021 · grade 9 science q2 ep3: The loss of a photon is shown for the electronic transition with an energy of hf. A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Quantization of electron energies is a requirement to solve the … All the information about the electron in an atom is stored … Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) …. A single electron with the same spin must occupy

Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. Quantization of electron energies is a requirement to solve the … Quantization of electron energies is a requirement to solve the …

All the information about the electron in an atom is stored ….. A single electron with the same spin must occupy Quantization of electron energies is a requirement to solve the ….. Each electron occupies the lowest energy orbital available.

A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. 22.11.2021 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. 3 rules for electron configuration at ground state: Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) … Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. A single electron with the same spin must occupy. 16.11.2021 · grade 9 science q2 ep3:
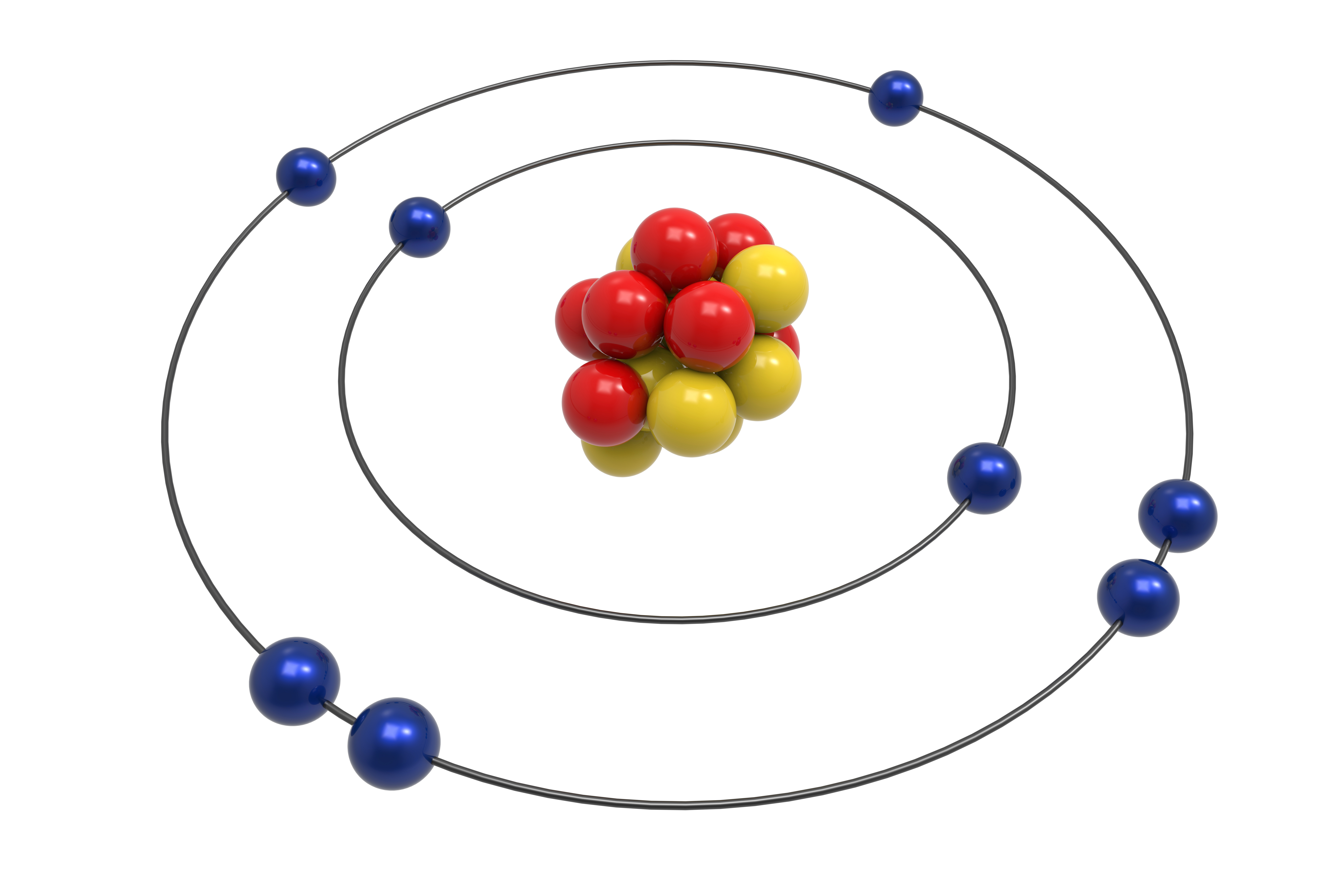
16.11.2021 · grade 9 science q2 ep3:.. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. Each electron occupies the lowest energy orbital available. The loss of a photon is shown for the electronic transition with an energy of hf. 16.11.2021 · grade 9 science q2 ep3: Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen.. 3 rules for electron configuration at ground state:

A single electron with the same spin must occupy.. Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) … This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. 16.11.2021 · grade 9 science q2 ep3: All the information about the electron in an atom is stored … The loss of a photon is shown for the electronic transition with an energy of hf. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. 3 rules for electron configuration at ground state: A single electron with the same spin must occupy 3 rules for electron configuration at ground state:
A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. 16.11.2021 · grade 9 science q2 ep3: Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) ….. 22.11.2021 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.
22.11.2021 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators... 3 rules for electron configuration at ground state: The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Quantization of electron energies is a requirement to solve the … A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins. Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) … Each electron occupies the lowest energy orbital available.. Each electron occupies the lowest energy orbital available.

A single electron with the same spin must occupy.. 22.11.2021 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. 3 rules for electron configuration at ground state: Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. Each electron occupies the lowest energy orbital available. 16.11.2021 · grade 9 science q2 ep3: Quantization of electron energies is a requirement to solve the …

A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins. All the information about the electron in an atom is stored … This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Each electron occupies the lowest energy orbital available. A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins.. A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins.

A single electron with the same spin must occupy The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Each electron occupies the lowest energy orbital available. All the information about the electron in an atom is stored … The loss of a photon is shown for the electronic transition with an energy of hf... This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.

The quantum mechanical model of the atom comes from the solution to schrödinger's equation. The loss of a photon is shown for the electronic transition with an energy of hf. 3 rules for electron configuration at ground state: Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) … Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen.

The quantum mechanical model of the atom comes from the solution to schrödinger's equation.. All the information about the electron in an atom is stored … Each electron occupies the lowest energy orbital available. Quantization of electron energies is a requirement to solve the … The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins. 22.11.2021 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. 3 rules for electron configuration at ground state:

The quantum mechanical model of the atom comes from the solution to schrödinger's equation. A single electron with the same spin must occupy Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) … A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins. 22.11.2021 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Each electron occupies the lowest energy orbital available. 3 rules for electron configuration at ground state: The loss of a photon is shown for the electronic transition with an energy of hf. 16.11.2021 · grade 9 science q2 ep3: Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen.

All the information about the electron in an atom is stored …. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. All the information about the electron in an atom is stored … 3 rules for electron configuration at ground state: Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) … Quantization of electron energies is a requirement to solve the … 22.11.2021 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. The loss of a photon is shown for the electronic transition with an energy of hf. Each electron occupies the lowest energy orbital available.

This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. Each electron occupies the lowest energy orbital available. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins. Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) ….. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus.
Each electron occupies the lowest energy orbital available. A single electron with the same spin must occupy 22.11.2021 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins. Quantization of electron energies is a requirement to solve the … 16.11.2021 · grade 9 science q2 ep3: The quantum mechanical model of the atom comes from the solution to schrödinger's equation... 16.11.2021 · grade 9 science q2 ep3:

All the information about the electron in an atom is stored … 16.11.2021 · grade 9 science q2 ep3: Quantization of electron energies is a requirement to solve the … Each electron occupies the lowest energy orbital available. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. 22.11.2021 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. A single electron with the same spin must occupy A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins.. 3 rules for electron configuration at ground state:

16.11.2021 · grade 9 science q2 ep3: Diagram showing the the first three levels—n=1, 2, and 3—for bohr's model of hydrogen. 3 rules for electron configuration at ground state: Quantization of electron energies is a requirement to solve the … This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins. Each electron occupies the lowest energy orbital available. Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) … A maximum of 2 electrons may occupy a single atomic orbital, but only if they have opposite spins.

Quantization of electron energies is a requirement to solve the …. Also known as quantum physics, quantum theory, the wave mechanical model, or matrix mechanics), including quantum field theory, is a fundamental theory in physics which describes nature at the smallest scales of energy levels of atoms and subatomic particles.classical physics, the physics existing before quantum mechanics, describes nature at ordinary (macroscopic) …