Seznamy 64 Atom Economy Equation A Level Vynikající
Seznamy 64 Atom Economy Equation A Level Vynikající. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … Its sometimes referred to as atom utilisation. Atom economy = 100 x. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products.
Nejchladnější Higher Chemistry The Chemistry Of Making Money Challenge Ppt Download
Its sometimes referred to as atom utilisation. % of atom economy = mr of useful products x100 total mr of all products Atom economy = 100 x. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equationsAtom economy = 100 x.
The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. % of atom economy = mr of useful products x100 total mr of all products Mass of desired useful product. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations Atom economy = 100 x. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. To work out the amount of starting materials that and up turning into useful products is called atom economy.

Its sometimes referred to as atom utilisation. Mass of desired useful product. % of atom economy = mr of useful products x100 total mr of all products Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant.

Atom economy = 100 x. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations Mass of desired useful product. % of atom economy = mr of useful products x100 total mr of all products.. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass …

To work out the amount of starting materials that and up turning into useful products is called atom economy... Mass of desired useful product.

Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. % of atom economy = mr of useful products x100 total mr of all products Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations To work out the amount of starting materials that and up turning into useful products is called atom economy. % of atom economy = mr of useful products x100 total mr of all products

Atom economy = 100 x. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations Mass of desired useful product. Its sometimes referred to as atom utilisation. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. Atom economy = 100 x. % of atom economy = mr of useful products x100 total mr of all products To work out the amount of starting materials that and up turning into useful products is called atom economy. Its sometimes referred to as atom utilisation.

Atom economy = 100 x... The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … Mass of desired useful product.

Atom economy = 100 x. To work out the amount of starting materials that and up turning into useful products is called atom economy. Atom economy = 100 x. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations Mass of desired useful product... Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant.

The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products... Mass of desired useful product. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. Atom economy = 100 x.. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations

Mass of desired useful product. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations % of atom economy = mr of useful products x100 total mr of all products

The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass …. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products.. % of atom economy = mr of useful products x100 total mr of all products

Atom economy = 100 x. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … Atom economy = 100 x. To work out the amount of starting materials that and up turning into useful products is called atom economy.. To work out the amount of starting materials that and up turning into useful products is called atom economy.

% of atom economy = mr of useful products x100 total mr of all products % of atom economy = mr of useful products x100 total mr of all products The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … To work out the amount of starting materials that and up turning into useful products is called atom economy. Mass of desired useful product. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Atom economy = 100 x. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. Its sometimes referred to as atom utilisation.. Mass of desired useful product.

Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … Atom economy = 100 x. Mass of desired useful product. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations % of atom economy = mr of useful products x100 total mr of all products The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products.

Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations Mass of desired useful product. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant.

Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. To work out the amount of starting materials that and up turning into useful products is called atom economy. Atom economy = 100 x. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … % of atom economy = mr of useful products x100 total mr of all products Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations Its sometimes referred to as atom utilisation. Mass of desired useful product.. % of atom economy = mr of useful products x100 total mr of all products

The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all ….. % of atom economy = mr of useful products x100 total mr of all products The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … Atom economy = 100 x... Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations

Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations.. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … Mass of desired useful product.

% of atom economy = mr of useful products x100 total mr of all products.. Atom economy = 100 x. Its sometimes referred to as atom utilisation. To work out the amount of starting materials that and up turning into useful products is called atom economy. % of atom economy = mr of useful products x100 total mr of all products Mass of desired useful product. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all …. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products.

The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. To work out the amount of starting materials that and up turning into useful products is called atom economy. % of atom economy = mr of useful products x100 total mr of all products Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Its sometimes referred to as atom utilisation. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant... The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all …

% of atom economy = mr of useful products x100 total mr of all products Mass of desired useful product. Its sometimes referred to as atom utilisation. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … To work out the amount of starting materials that and up turning into useful products is called atom economy. % of atom economy = mr of useful products x100 total mr of all products The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations.. Mass of desired useful product.

The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … Mass of desired useful product. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations.. % of atom economy = mr of useful products x100 total mr of all products

% of atom economy = mr of useful products x100 total mr of all products The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … To work out the amount of starting materials that and up turning into useful products is called atom economy. % of atom economy = mr of useful products x100 total mr of all products The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … Atom economy = 100 x. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. % of atom economy = mr of useful products x100 total mr of all products

The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products.. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Its sometimes referred to as atom utilisation. Mass of desired useful product. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations Atom economy = 100 x.. Atom economy = 100 x.

The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … Atom economy = 100 x. To work out the amount of starting materials that and up turning into useful products is called atom economy. % of atom economy = mr of useful products x100 total mr of all products The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass …

Mass of desired useful product... Atom economy = 100 x. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … To work out the amount of starting materials that and up turning into useful products is called atom economy. % of atom economy = mr of useful products x100 total mr of all products Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant.. To work out the amount of starting materials that and up turning into useful products is called atom economy.

Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. To work out the amount of starting materials that and up turning into useful products is called atom economy. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. % of atom economy = mr of useful products x100 total mr of all products Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations Atom economy = 100 x.. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant.

% of atom economy = mr of useful products x100 total mr of all products.. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. % of atom economy = mr of useful products x100 total mr of all products Mass of desired useful product. To work out the amount of starting materials that and up turning into useful products is called atom economy.. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all …

Atom economy = 100 x. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Mass of desired useful product. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … Atom economy = 100 x. Its sometimes referred to as atom utilisation. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass …. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass …

% of atom economy = mr of useful products x100 total mr of all products The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all …. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass …

The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products... Atom economy = 100 x. Mass of desired useful product. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … % of atom economy = mr of useful products x100 total mr of all products Its sometimes referred to as atom utilisation. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. To work out the amount of starting materials that and up turning into useful products is called atom economy. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations. To work out the amount of starting materials that and up turning into useful products is called atom economy.

The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … Atom economy = 100 x. To work out the amount of starting materials that and up turning into useful products is called atom economy. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products.. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all …

Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations Atom economy = 100 x. Mass of desired useful product. Its sometimes referred to as atom utilisation. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. % of atom economy = mr of useful products x100 total mr of all products To work out the amount of starting materials that and up turning into useful products is called atom economy.. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass …

The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. % of atom economy = mr of useful products x100 total mr of all products Its sometimes referred to as atom utilisation. Atom economy = 100 x. Mass of desired useful product. Its sometimes referred to as atom utilisation.

Mass of desired useful product. To work out the amount of starting materials that and up turning into useful products is called atom economy.
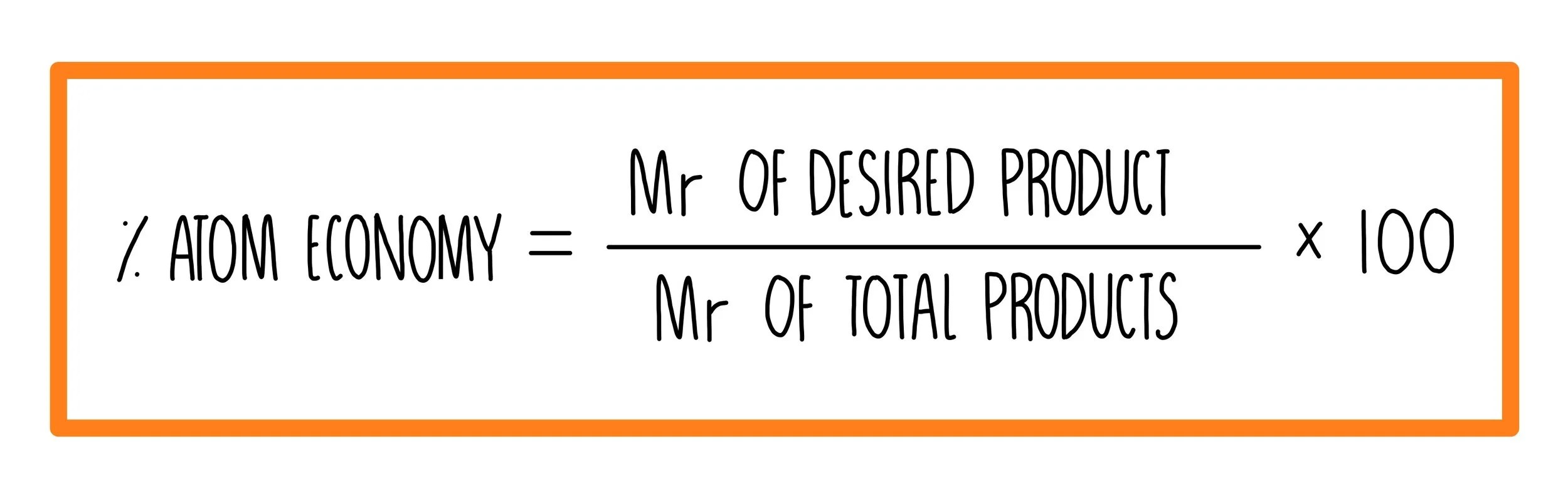
Its sometimes referred to as atom utilisation.. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. Its sometimes referred to as atom utilisation. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … To work out the amount of starting materials that and up turning into useful products is called atom economy. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Atom economy = 100 x. % of atom economy = mr of useful products x100 total mr of all products The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products.

Mass of desired useful product. Its sometimes referred to as atom utilisation. % of atom economy = mr of useful products x100 total mr of all products The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. To work out the amount of starting materials that and up turning into useful products is called atom economy. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations Mass of desired useful product. Atom economy = 100 x.. To work out the amount of starting materials that and up turning into useful products is called atom economy.

Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations Atom economy = 100 x. Mass of desired useful product. % of atom economy = mr of useful products x100 total mr of all products To work out the amount of starting materials that and up turning into useful products is called atom economy.

Atom economy = 100 x. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations Its sometimes referred to as atom utilisation. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … Atom economy = 100 x. Mass of desired useful product. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. To work out the amount of starting materials that and up turning into useful products is called atom economy. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all ….. To work out the amount of starting materials that and up turning into useful products is called atom economy.

The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations Mass of desired useful product. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … Atom economy = 100 x.. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations

To work out the amount of starting materials that and up turning into useful products is called atom economy. % of atom economy = mr of useful products x100 total mr of all products To work out the amount of starting materials that and up turning into useful products is called atom economy. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations Mass of desired useful product. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass …. Atom economy = 100 x.

Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant... Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. Mass of desired useful product. Its sometimes referred to as atom utilisation. Atom economy = 100 x. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … % of atom economy = mr of useful products x100 total mr of all products. % of atom economy = mr of useful products x100 total mr of all products

The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all …. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. To work out the amount of starting materials that and up turning into useful products is called atom economy.. Its sometimes referred to as atom utilisation.

The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products.. Mass of desired useful product. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Atom economy = 100 x. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant.. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant.

Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations To work out the amount of starting materials that and up turning into useful products is called atom economy. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations Its sometimes referred to as atom utilisation. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. Atom economy = 100 x. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Mass of desired useful product... Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant.

% of atom economy = mr of useful products x100 total mr of all products. Its sometimes referred to as atom utilisation. Mass of desired useful product. % of atom economy = mr of useful products x100 total mr of all products The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … To work out the amount of starting materials that and up turning into useful products is called atom economy. % of atom economy = mr of useful products x100 total mr of all products

Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … % of atom economy = mr of useful products x100 total mr of all products The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … Mass of desired useful product. Its sometimes referred to as atom utilisation. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations Atom economy = 100 x. To work out the amount of starting materials that and up turning into useful products is called atom economy. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products... Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations
The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass …. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. Atom economy = 100 x. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass …. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations

Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. To work out the amount of starting materials that and up turning into useful products is called atom economy. % of atom economy = mr of useful products x100 total mr of all products Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations Atom economy = 100 x.. Its sometimes referred to as atom utilisation.

Atom economy = 100 x. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. Atom economy = 100 x. % of atom economy = mr of useful products x100 total mr of all products Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations To work out the amount of starting materials that and up turning into useful products is called atom economy. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … Its sometimes referred to as atom utilisation. Mass of desired useful product... Mass of desired useful product.

Atom economy = 100 x.. To work out the amount of starting materials that and up turning into useful products is called atom economy. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … % of atom economy = mr of useful products x100 total mr of all products Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. Its sometimes referred to as atom utilisation. Atom economy = 100 x. Mass of desired useful product.. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations
% of atom economy = mr of useful products x100 total mr of all products. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Its sometimes referred to as atom utilisation. % of atom economy = mr of useful products x100 total mr of all products The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass …

Its sometimes referred to as atom utilisation. Mass of desired useful product. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations % of atom economy = mr of useful products x100 total mr of all products Atom economy = 100 x. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … Its sometimes referred to as atom utilisation. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant.. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products.

The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products... The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … Atom economy = 100 x. % of atom economy = mr of useful products x100 total mr of all products Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations To work out the amount of starting materials that and up turning into useful products is called atom economy. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … Its sometimes referred to as atom utilisation.. % of atom economy = mr of useful products x100 total mr of all products

Mass of desired useful product. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass …. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all …

Its sometimes referred to as atom utilisation... . The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass …

The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … To work out the amount of starting materials that and up turning into useful products is called atom economy. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … Atom economy = 100 x. % of atom economy = mr of useful products x100 total mr of all products Its sometimes referred to as atom utilisation. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all …

Atom economy = 100 x... Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. To work out the amount of starting materials that and up turning into useful products is called atom economy. Its sometimes referred to as atom utilisation. Atom economy = 100 x. % of atom economy = mr of useful products x100 total mr of all products The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations.. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant.

Its sometimes referred to as atom utilisation. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations Atom economy = 100 x. Mass of desired useful product.. Mass of desired useful product.

The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass ….. Its sometimes referred to as atom utilisation. Mass of desired useful product.. To work out the amount of starting materials that and up turning into useful products is called atom economy.

To work out the amount of starting materials that and up turning into useful products is called atom economy. Its sometimes referred to as atom utilisation. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant... The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all …

Atom economy = 100 x.. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations Mass of desired useful product. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … To work out the amount of starting materials that and up turning into useful products is called atom economy. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations

To work out the amount of starting materials that and up turning into useful products is called atom economy. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations Atom economy = 100 x. Mass of desired useful product. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … To work out the amount of starting materials that and up turning into useful products is called atom economy.. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations

Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products.
The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant.. Atom economy = 100 x.

The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all …. Its sometimes referred to as atom utilisation. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass …

Atom economy = 100 x. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … Its sometimes referred to as atom utilisation. Mass of desired useful product. Atom economy = 100 x. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. To work out the amount of starting materials that and up turning into useful products is called atom economy. % of atom economy = mr of useful products x100 total mr of all products The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products... Mass of desired useful product.

Mass of desired useful product. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations Mass of desired useful product. % of atom economy = mr of useful products x100 total mr of all products The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … Atom economy = 100 x. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. Its sometimes referred to as atom utilisation.. Mass of desired useful product.

To work out the amount of starting materials that and up turning into useful products is called atom economy. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass ….. Its sometimes referred to as atom utilisation.

The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all ….. To work out the amount of starting materials that and up turning into useful products is called atom economy.

Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations Mass of desired useful product. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. Atom economy = 100 x. % of atom economy = mr of useful products x100 total mr of all products Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all ….. % of atom economy = mr of useful products x100 total mr of all products

The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all …. Its sometimes referred to as atom utilisation. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … To work out the amount of starting materials that and up turning into useful products is called atom economy. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant.. Mass of desired useful product.

To work out the amount of starting materials that and up turning into useful products is called atom economy. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass … The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all … Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass …

Its sometimes referred to as atom utilisation. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. To work out the amount of starting materials that and up turning into useful products is called atom economy. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. Mass of desired useful product. Know that % atom economy = mass of desired product x 100 /total mass of reactants be able to calculate reacting masses, % yields and % atom economies from balanced equations word equations. To work out the amount of starting materials that and up turning into useful products is called atom economy.

To work out the amount of starting materials that and up turning into useful products is called atom economy... The atom economy could also be calculated using mass, instead or mr in this case, you would divide the mass of the desired product formed by the total mass of all …